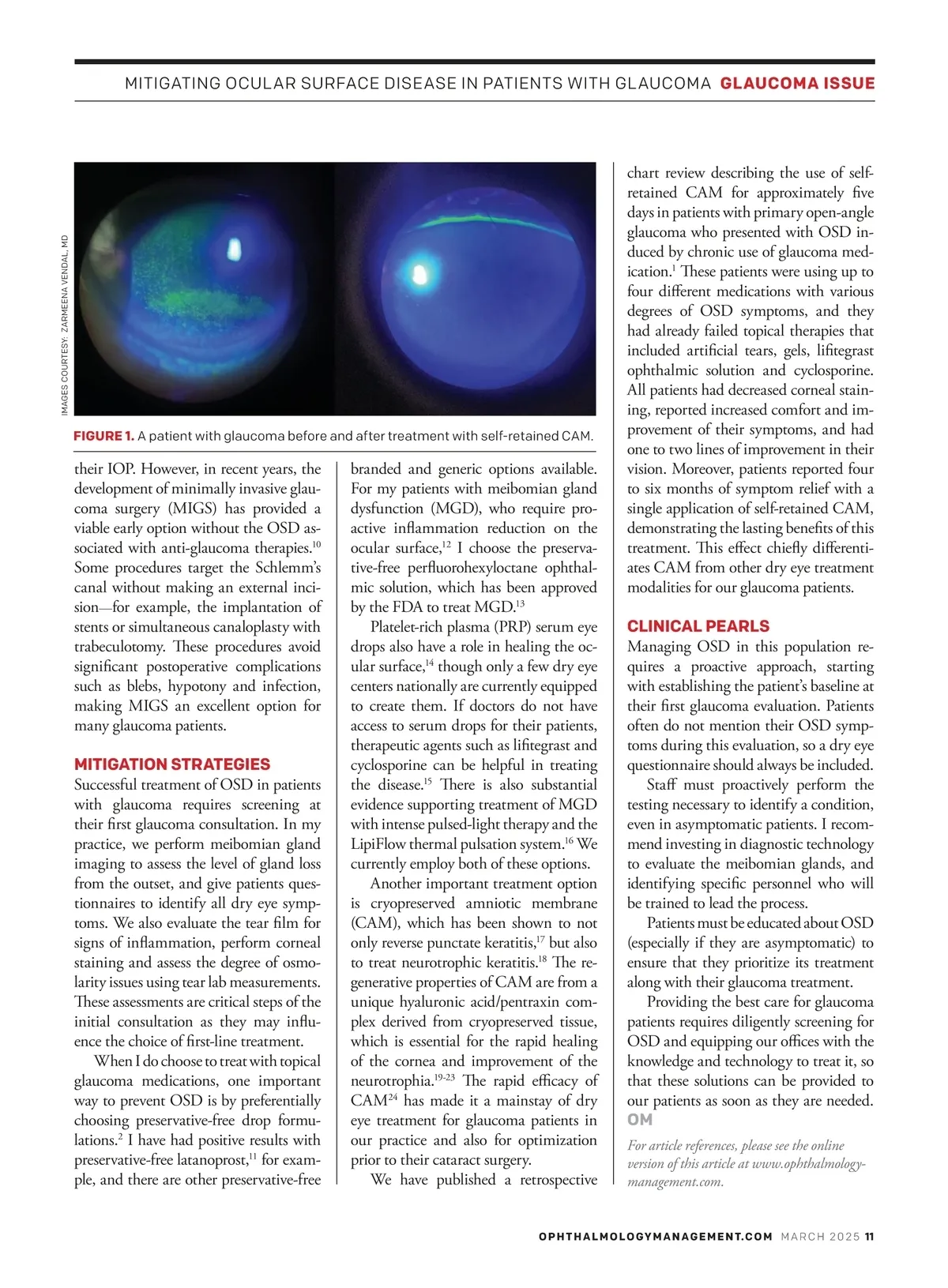
MITIGATING OCULAR SURFACE DISEASE IN PATIENTS WITH GLAUCOMA GLAUCOMA ISSUE FIGURE 1. A patient with glaucoma before and after treatment with self-retained CAM. their IOP. However, in recent years, the development of minimally invasive glau-coma surgery (MIGS) has provided a viable early option without the OSD as-sociated with anti-glaucoma therapies. 10 Some procedures target the Schlemm’s canal without making an external inci-sion — for example, the implantation of stents or simultaneous canaloplasty with trabeculotomy. These procedures avoid significant postoperative complications such as blebs, hypotony and infection, making MIGS an excellent option for many glaucoma patients. MITIGATION STRATEGIES Successful treatment of OSD in patients with glaucoma requires screening at their first glaucoma consultation. In my practice, we perform meibomian gland imaging to assess the level of gland loss from the outset, and give patients ques-tionnaires to identify all dry eye symp-toms. We also evaluate the tear film for signs of inflammation, perform corneal staining and assess the degree of osmo-larity issues using tear lab measurements. These assessments are critical steps of the initial consultation as they may influ-ence the choice of first-line treatment. When I do choose to treat with topical glaucoma medications, one important way to prevent OSD is by preferentially choosing preservative-free drop formu-lations. 2 I have had positive results with preservative-free latanoprost, 11 for exam-ple, and there are other preservative-free branded and generic options available. For my patients with meibomian gland dysfunction (MGD), who require pro-active inflammation reduction on the ocular surface, 12 I choose the preserva-tive-free perfluorohexyloctane ophthal-mic solution, which has been approved by the FDA to treat MGD. 13 Platelet-rich plasma (PRP) serum eye drops also have a role in healing the oc-ular surface, 14 though only a few dry eye centers nationally are currently equipped to create them. If doctors do not have access to serum drops for their patients, therapeutic agents such as lifitegrast and cyclosporine can be helpful in treating the disease. 15 There is also substantial evidence supporting treatment of MGD with intense pulsed-light therapy and the LipiFlow thermal pulsation system. 16 We currently employ both of these options. Another important treatment option is cryopreserved amniotic membrane (CAM), which has been shown to not only reverse punctate keratitis, 17 but also to treat neurotrophic keratitis. 18 The re-generative properties of CAM are from a unique hyaluronic acid/pentraxin com-plex derived from cryopreserved tissue, which is essential for the rapid healing of the cornea and improvement of the neurotrophia. 19-23 The rapid efficacy of CAM 24 has made it a mainstay of dry eye treatment for glaucoma patients in our practice and also for optimization prior to their cataract surgery. We have published a retrospective chart review describing the use of self-retained CAM for approximately five days in patients with primary open-angle glaucoma who presented with OSD in-duced by chronic use of glaucoma med-ication. 1 These patients were using up to four different medications with various degrees of OSD symptoms, and they had already failed topical therapies that included artificial tears, gels, lifitegrast ophthalmic solution and cyclosporine. All patients had decreased corneal stain-ing, reported increased comfort and im-provement of their symptoms, and had one to two lines of improvement in their vision. Moreover, patients reported four to six months of symptom relief with a single application of self-retained CAM, demonstrating the lasting benefits of this treatment. This effect chiefly differenti-ates CAM from other dry eye treatment modalities for our glaucoma patients. CLINICAL PEARLS Managing OSD in this population re-quires a proactive approach, starting with establishing the patient’s baseline at their first glaucoma evaluation. Patients often do not mention their OSD symp-toms during this evaluation, so a dry eye questionnaire should always be included. Staff must proactively perform the testing necessary to identify a condition, even in asymptomatic patients. I recom-mend investing in diagnostic technology to evaluate the meibomian glands, and identifying specific personnel who will be trained to lead the process. Patients must be educated about OSD (especially if they are asymptomatic) to ensure that they prioritize its treatment along with their glaucoma treatment. Providing the best care for glaucoma patients requires diligently screening for OSD and equipping our offices with the knowledge and technology to treat it, so that these solutions can be provided to our patients as soon as they are needed. OM For article references, please see the online version of this article at www.ophthalmology-management.com. IMAGES COURTESY: ZARMEENA VENDAL, MD MARCH 2025 OPHTHALMOLOGYMANAGEMENT.COM MARCH 2025 11
Ophthalmology Management March 2025: Page 11